Aresus Pharma GmbH
Kastanienallee 46
15344 Strausberg
Phone: +49 (0) 3341 589 9040
Fax: +49 (0) 3341 589 9041
E-Mail: info@aresus.de
For scientific and study-related questions please contact: clinical@aresus.de
Phone.: 089/997 42 82 71

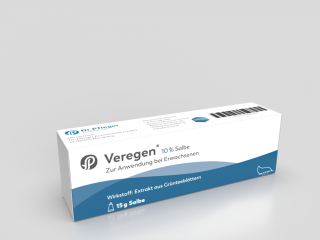
Aresus took over Veregen® from Medigene (see Press Release)incl. all patents, licenses, know-how as well as rights to use the brand name, the Drug Master File (DMF) of the active ingredient and the inventory of the active pharmaceutical ingredient.

Aresus Pharma GmbH
Kastanienallee 46
15344 Strausberg
Phone: +49 (0) 3341 589 9040
Fax: +49 (0) 3341 589 9041
E-Mail: info@aresus.de
For scientific and study-related questions please contact: clinical@aresus.de
Phone.: 089/997 42 82 71
Veregen® is the first drug in its class for the local treatment of genital warts and is based on a defined extract of green tea leaves produced in a complex and specifically developed process. Veregen® was the first and is one of the few innovative herbal medicines which was approved by the U.S. Food and Drug Administration (FDA).